Scripps Research scientists break down the basics of SARS-CoV-2
One of the great endeavors of research is to take what we know about the tiniest molecular machines and translate that into medicines that go on to change the world. As we continue to experience the personal and societal e2ects of the COVID-19pandemic, scientists are unearthing the tricks of the SARS-CoV-2virus and developing thoughtful ways to counteract it. Whether it’s a precise, nanoparticle vaccine or simply scrubbing our hands, any protective measures depend on the fascinating science of this new pathogen.
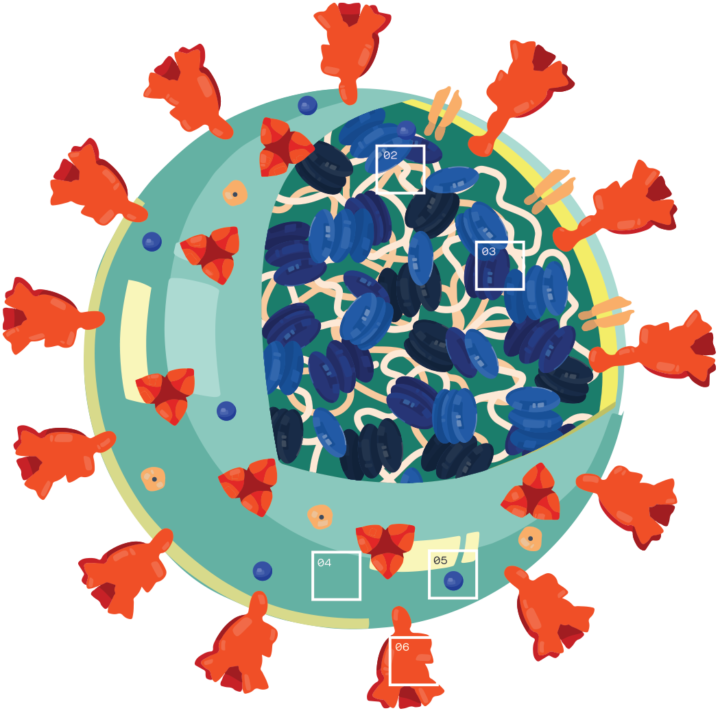
I. What are the parts of a coronavirus?
Coronaviruses are a large family of viruses, some of which infect humans.
01 – The coronavirus at the root of COVID-19 is the newest known member of this family. And like other coronaviruses that infect people, the new coronavirus causes respiratory disease, among other symptoms.
02 – At their core, coronaviruses contain a genetic blueprint called RNA (beige), similar to DNA. The single-stranded RNA acts as a molecular message that enables production of proteins needed for other elements of the virus.
03 – Bound to this string of RNA are nucleoproteins (dark blue discs) —proteins that help give the virus its structure and enable it to replicate.
04 – Encapsulating the RNA genome is the viral envelope (teal), which protects the virus when it is outside of a host cell. This outer envelope is made from a layer of lipids, a waxy barrier containing fat molecules. As well as protecting the precious genetic cargo, this layer anchors the different structural proteins needed by the virus to infect cells.
05 – Envelope proteins (dark blue dots) embedded in this layer aid the assembly of new virus particles once it has infected a cell.
06 – The bulbous projections seen on the outside of the coronavirus are spike proteins (red-orange). This fringe of proteins gives the virus its crown or halo-like appearance under the microscope, from which the Latin name corona is derived. The spike proteins act as grappling hooks that allow the virus to latch onto host cells and crack them open for infection. Like all viruses, coronaviruses are unable to thrive and reproduce outside of a living host.
II. How does the novel coronavirus infect a cell?
Due to its unique features, the novel coronavirus is particularly good at infecting new cells, both in the upper respiratory tract, as well as deeper down in the lungs. Here’s a look at how the process takes place.
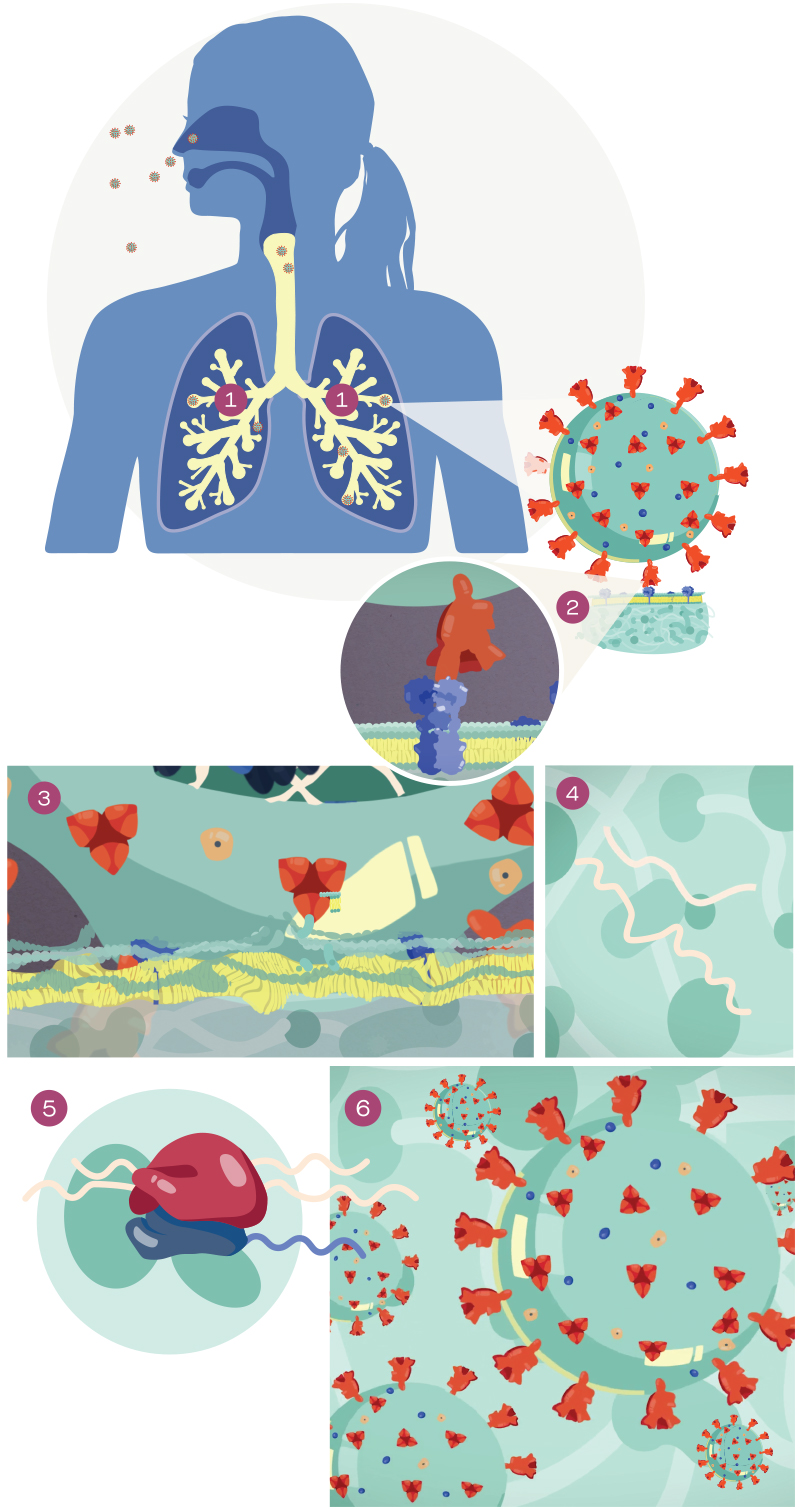
01 – The microscopic virus enters through the nose or mouth, where it begins its infection of our airways.
02 – The outer spike protein of the coronavirus latches onto specific receptors on the surface of cells in our respiratory tract. In the case of COVID-19, the virus latches on to the ACE2 receptor.
03 – This binding triggers the process by which the virus fuses into human cells. The viral envelope merges with the oily membrane of our own cells, allowing the virus to release its genetic material into the inside of the healthy cell.
04 – The genetic blueprint of the virus is RNA (instead of DNA), which acts as a molecular message, instructing our host cell machinery to read the template and translate it into proteins that make up new virus particles.
05 – The hijacking persists, as the human host cell continues to generate more copies of the virus, assemble these copies into viable particles and traffic them to the outer edges of the cell for release.
06 – Each infected cell may produce and release millions of copies of the virus, which can then go on to infect other neighboring cells, as well as neighboring people when they are expelled from the airways in droplets via coughing and sneezing.
III. How do antibodies stop the coronavirus?

Antibodies are Y-shaped proteins produced by our immune system after we are exposed to a disease-causing invader, such as bacteria or a virus. These proteins help the body remember the threat and be better prepared for the next encounter.
Antibodies to the novel coronavirus recognize the virus’s outer spike protein enabling them to bind to the surface of the virus, preventing the virus particles from attaching to the host cell.
Even if the virus successfully attaches, antibodies can prevent it from penetrating our cell membranes. If the virus does manage to enter the cell, antibodies can still prevent the virus from releasing its genome into the host cell for replication.
Once antibodies neutralize the virus, they can target the virus for destruction by signaling to other components of our immune system.
Scientists are trying to use antibodies both as a way to treat COVID-19 and prevent the disease from taking hold. Some researchers are studying antibodies taken from recovered COVID-19 patients. These antibodies could act as a medicine for the newly infected.
Other scientists are focused on how the antibody interacts with coronavirus spike proteins, which may enable them to design a successful vaccine that instructs our bodies to generate effective antibodies against the virus.
The long-term hope is that we might also be able to identify “broadly neutralizing antibodies,” a type of antibody that could defuse not just this specific viral strain, but also offshoots that occur because of natural mutations in the virus over time.
IV. How would potential antiviral drugs work against the coronavirus?
In contrast to a preventative vaccine, most antiviral medicines would target the virus in people who have already been infected. These drugs can work in several different ways, targeting various aspects of the virus particle or even the human host cell that the virus invades.

Some antiviral drugs in development disarm the surface spike proteins of the virus, preventing the virus from latching onto the host cell. Others are designed to stop the virus from replicating after it enters the host cell. A number of these molecules block key viral components called proteases; this prevents key viral proteins from maturing and copying the virus’s genetic material. Drugs (such as remdesivir) may also interfere with this copying process by tricking the virus into merging the drug molecules into its RNA backbone, which disrupts the genetic blueprint and prevents replication. Alternatively, the viral production line could be halted by modifying elements of host cells. Certain treatments may interact with receptors on the inside of our cells that act like switches, and which are often manipulated by the virus to aid infection and replication.
Just as completely novel drugs are being developed for the coronavirus, scientists are also screening treatments that are already approved for other diseases—as many of these drug molecules exhibit antiviral properties. Because existing drugs are already known to be safe in humans, they could be developed into a viable COVID-19 treatment faster than new drugs that have never been tested in people. Researchers are also testing existing drugs that could be used in combination with each other; this approach reduces the dose needed for each drug and helps to minimize side effects for patients.
V. How would a vaccine work against COVID-19?
After we have been exposed to an infection, our immune system remembers the threat by producing antibodies—proteins that can prepare the body’s defenses for the next encounter. Vaccines mimic this process, encouraging the immune system to make antibodies that could quickly recognize and disable the invader upon contact, thereby preventing or minimizing illness.
Scientific studies have shown that human antibodies to the novel coronavirus are able to recognize the outer spike protein of the virus. These discoveries are driving several different approaches to design vaccines that could protect against COVID-19.
A traditional approach to vaccine development involves testing weakened or inactivated forms of the SARS-CoV-2 virus to determine whether they can still prompt an antibody response.
Alternatively, some researchers are studying whether it’s possible to use small pieces of the virus, such as isolated spike proteins, to achieve the desired immune response. This approach is known as a subunit vaccine.
Antibodies themselves are also being investigated as clues for how to design a vaccine, particularly antibodies from recent survivors of COVID-19 or those produced by individuals in response to the closely related SARS virus from the outbreak that began in 2002.
Recently, labs are also exploring gene-based vaccines, an approach that incorporates parts of the virus’s genetic blueprint into isolated host cells. These cells are then instructed to make the features of the virus that circulating antibodies might recognize.
Once a potential vaccine is discovered, a number of checkpoints exist before it can be administered to people. First are preclinical tests, which involve experiments in a laboratory and with animals. Scientists must ensure the vaccine candidate is not only effective, but also safe. For example, an antibody response to an imperfect vaccine could, under very rare circumstances, end up increasing the danger of becoming infected.
When the potential vaccine achieves the necessary preclinical results, clinical trials can begin in a small group of people. As the vaccine candidate advances, it is tested on increasing numbers of people, with scientists and doctors closely monitoring safety, efficacy and dosing. Upon successful completion of clinical trials, the vaccine candidate must be reviewed and approved by regulatory agencies such as the FDA before large-scale manufacturing and distribution gets underway and the licensed vaccine is administered widely.
VI. How could anti-inflammatory drugs work against the coronavirus?

The human immune system is designed to stop diseases before they make us sick. This occurs via highly controlled inflammatory and immune reactions. But, in some severe infections, the immune system can dangerously overreact—inflicting damage on the healthy surrounding tissues.
In patients with COVID-19, a severe immune reaction in the lungs can cause inflammation that leads to acute respiratory distress. Several ongoing clinical trials are testing drugs that may be able to prevent this dangerous inflammation in the lungs or other organs. Many of these therapies are already used to treat other diseases, where they help to temper the harmful effects of the immune system by blocking specific inflammatory signals.
VII. How do soap and hand sanitizer work against COVID-19?
The coronavirus that causes COVID-19 can be easily destroyed by soap. But why? Soap molecules consist of long molecules called surfactants that contain a head and a tail. While the head prefers to bond with water, the tail prefers oils and fats instead. Trying to escape water, the tails bury themselves into the fatty outer layer of the virus. The soap is able to break apart the chemical bonds of this vulnerable viral envelope, forcing it to break open and spill its contents, rendering it useless. However, to do its job effectively, the soap needs to reach all the creases of our hands and requires some time to interact with the virus. This is why we should spend at least 20 seconds washing our hands. Alcohol-based hand sanitizers work in a similar way, as alcohol can destroy the essential envelope proteins that surround the virus. Hand sanitizer needs to be at least 60% alcohol to achieve this, and the approach is still less effective than diligently washing hands with soap and hot water.
