
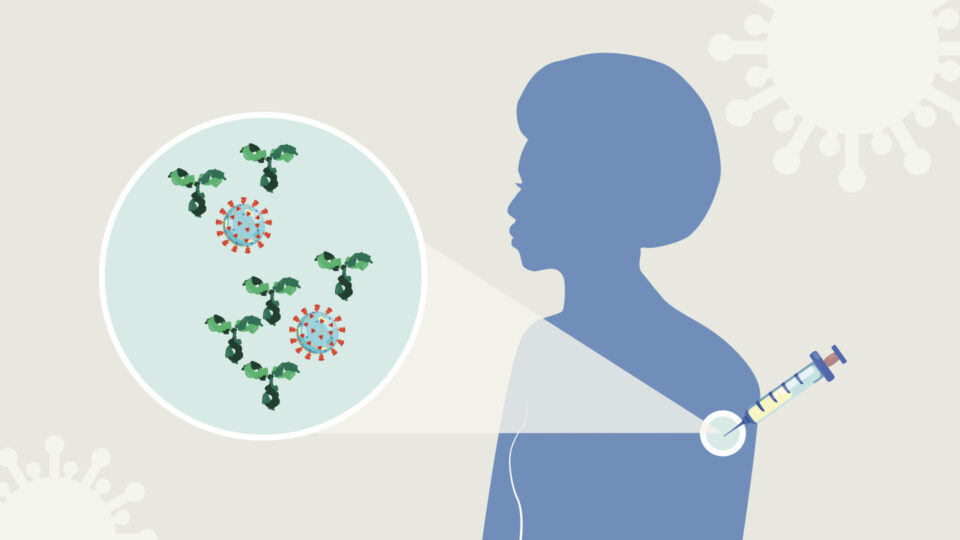
I. How does a vaccine work against COVID-19?
After we have been exposed to an infection, our immune system remembers the threat, in particular by producing antibodies. These are proteins that circulate in the blood and throughout the body; they quickly recognize and disable the invader upon contact, thereby preventing or minimizing illness. This is why we usually do not get sick with the same bug twice; we are immune. Vaccines mimic this process, encouraging the immune system to make antibodies without us having to go through the illness.
Once a potential vaccine is discovered, a number of checkpoints exist before it can be administered to people. First are preclinical tests, which involve experiments in a laboratory and with animals. Scientists must ensure the vaccine candidate is not only effective, but also safe. For example, an antibody response to an imperfect vaccine could, under extremely rare circumstances, end up increasing the danger of becoming infected.
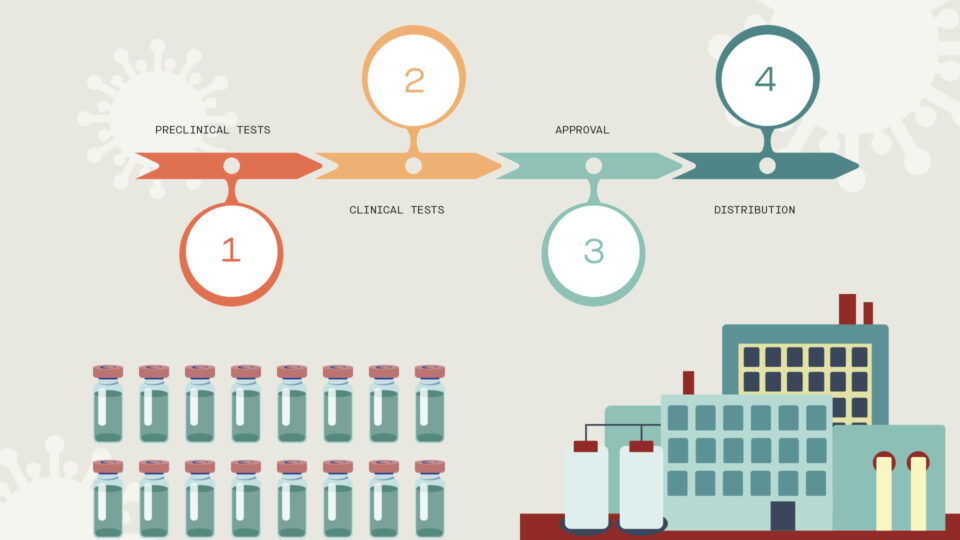
When the potential vaccine achieves the necessary preclinical results, clinical trials can begin in a small group of people. As the vaccine candidate advances, it is tested on increasing numbers of people, with scientists and doctors closely monitoring safety, efficacy and dosing. Upon successful completion of clinical trials, the vaccine candidate must be reviewed and approved by regulatory agencies such as the FDA before large-scale manufacturing and distribution gets underway and the licensed vaccine is administered widely.
II. Science Simplified: COVID-19 mutations give rise to new variants
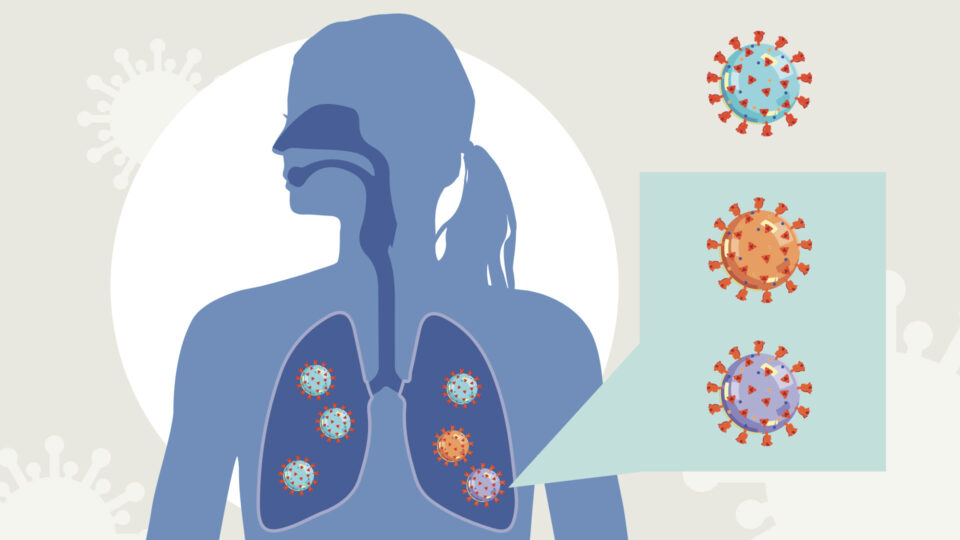
As the virus behind the COVID-19 pandemic, SARS-CoV-2, spreads and infects millions of people, it is evolving new characteristics and adding new branches to its family tree. But how does this happen and what are the implications?
Mutations are a common occurrence in most viruses. As a virus replicates inside a host, random errors while copying its genetic code can lead to mutations. Much of the time, these mistakes can be harmless or make a virus weaker. But occasionally, these mutations can help the virus to thrive, giving rise to new variants, which people also refer to as “strains.” Just as evolution would have it, those variants with the most adaptive features will be the ones that survive and multiply, which can lead to multiple variants of a given virus within a population.
Once a successful new variant emerges, there could be a number of potential consequences. The mutations could help the virus’s ability to infect the host and spread from person to person. They could also change the severity of the disease after infection, or make the virus harder to detect by existing diagnostic tests. In addition, mutations could enable that variant of the virus to be protected against antiviral medicines or vaccines.
Many COVID-19 variants are circulating throughout the world, such as those identified in South Africa and Brazil. One of the most notable variants emerged from the United Kingdom and has quickly spread across continents. Known as B.1.1.7, this variant has an unusually large number of mutations, with many of them in the spike protein, which the virus uses to attach to and infect host cells.
Research suggests that the new variant is associated with increased transmission, meaning the virus could be more contagious. Scientists are still studying the variant’s severity for those infected, and the impact of the mutations on vaccine efficacy is still to be fully determined. This continued “strain surveillance” is critical for monitoring the course of a pandemic over time and determining the most effective public health response.
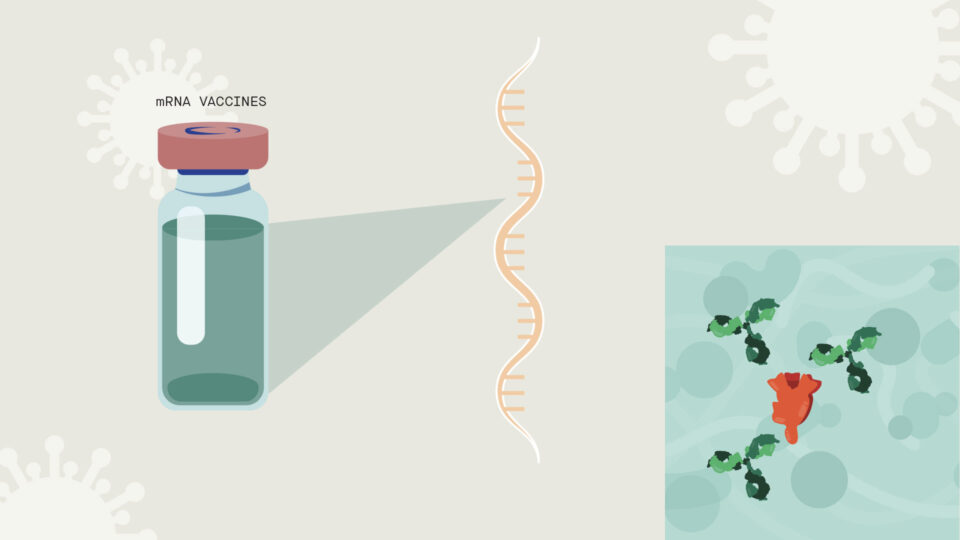
Some of the leading SARS-CoV-2 vaccine candidates are “mRNA vaccines,” based on incorporating the genetic blueprint for the key spike protein on the virus surface into a formula that when injected into humans instructs our own cells to make the spike protein. In turn, the body then makes antibodies against the spike protein and they protect us against viral infection.
This strategy is faster than more traditional approaches, which often involve generating weakened or inactivated forms of a live virus or making large amounts of the spike protein to determine whether they can prompt an antibody response.